The cruise is focusing on measuring iron in the water column and sediments. Iron concentration in sediments is very high (milli molar-to-1%). However, the range of concentrations of iron in the water column varies from pico-to-nano molar concentration – similar to finding a green lentil within 1 billion (nanomolar) to 1 trillion of red lentils (picomolar)!
Because of the large differences between these two environments, we have to be extra careful when sampling and working on the ship. To sample the water column, we use an ultra clean CTD rosette that measures Conductivity Temperature, and Depth (CTD). This rosette was specially made by NIOZ technicians for low concentration sampling. For more details, please see our blog from last year on the Black Sea: http://geoblog.weebly.com/expedition-black-sea-2015.
To sample and work with these precious samples, we wear special clothing to avoid contact with iron-plentiful materials such as dusts, fluff from clothes, hair, etc.. . Our special clothes look at lot like we are working in a bakery including the hair nets. We are very fashionable!
Then start the party! Once the CTD is within the clean container (specially designed to deliver clean air, without particles that could contaminate our samples), we start to sample the water from the different bottles (that sampled different depths) of the clean CTD.
Because of the large differences between these two environments, we have to be extra careful when sampling and working on the ship. To sample the water column, we use an ultra clean CTD rosette that measures Conductivity Temperature, and Depth (CTD). This rosette was specially made by NIOZ technicians for low concentration sampling. For more details, please see our blog from last year on the Black Sea: http://geoblog.weebly.com/expedition-black-sea-2015.
To sample and work with these precious samples, we wear special clothing to avoid contact with iron-plentiful materials such as dusts, fluff from clothes, hair, etc.. . Our special clothes look at lot like we are working in a bakery including the hair nets. We are very fashionable!
Then start the party! Once the CTD is within the clean container (specially designed to deliver clean air, without particles that could contaminate our samples), we start to sample the water from the different bottles (that sampled different depths) of the clean CTD.
The first set of samples to be taken is for alkalinity, pH, DOC and nutrients. Mathilde samples for alkalinity, pH and dissolved inorganic carbon (DIC) while Sharyn collects for nutrients. The instruments in Sharyn’s lab/container measure nitrate, nitrite, ammonia, phosphate, and silica- these are key nutrients for phytoplankton (the little organisms that build the base of the marine food web). By measuring these parameters, the concentrations of biological transformations (phytoplankton being productive or consumers eating up the plankton and releasing CO2) help to show the zonations of different processes within the water column like oxygen present, low oxygen, or anoxic.
After nutrients are collected, the trace metal team starts to collect for future analysis in our university labs. For each CTD bottle, we collect a series of different types of samples by using various filters. The sampling procedure follows a rigorous and consistent protocol to minimize contamination. We fill the bottle once with few milliliters (mL) of the sample to be collected, shake the bottle for few seconds to make sure that the sample covers and rinses the wall of the bottle, pour of the sample into the cap and empty the cap onto the thread of the bottle. We repeat this step three times before filling the bottle with the sample. Moreover, every time we use a capsule filter, we pass at least a liter through it to make sure that the sample that we collect is representative of the actual sample, it is a conditioning step basically.
The first type of samples to be collected is unfiltered seawater (to determine total concentrations of trace metals such as iron, manganese, zinc, cobalt, aluminium, etc..). Then we collect filtered samples using 0.2 µm size pore filter. They are small capsules that collect all particles and plankton greater than 0.2 µm, while anything smaller will pass through the filter to the bottle and will represent our 0.2 µm filtered fraction of trace metals in seawater. Finally, we collect samples on 0.02 um using large capsules.
The process of filling these bottles can be very long task, especially when there is many bottles needing filtration or if the bottles are of a large volume (1 liter filtered is very slow). Organisation in the sampling strategy becomes really important!
After nutrients are collected, the trace metal team starts to collect for future analysis in our university labs. For each CTD bottle, we collect a series of different types of samples by using various filters. The sampling procedure follows a rigorous and consistent protocol to minimize contamination. We fill the bottle once with few milliliters (mL) of the sample to be collected, shake the bottle for few seconds to make sure that the sample covers and rinses the wall of the bottle, pour of the sample into the cap and empty the cap onto the thread of the bottle. We repeat this step three times before filling the bottle with the sample. Moreover, every time we use a capsule filter, we pass at least a liter through it to make sure that the sample that we collect is representative of the actual sample, it is a conditioning step basically.
The first type of samples to be collected is unfiltered seawater (to determine total concentrations of trace metals such as iron, manganese, zinc, cobalt, aluminium, etc..). Then we collect filtered samples using 0.2 µm size pore filter. They are small capsules that collect all particles and plankton greater than 0.2 µm, while anything smaller will pass through the filter to the bottle and will represent our 0.2 µm filtered fraction of trace metals in seawater. Finally, we collect samples on 0.02 um using large capsules.
The process of filling these bottles can be very long task, especially when there is many bottles needing filtration or if the bottles are of a large volume (1 liter filtered is very slow). Organisation in the sampling strategy becomes really important!
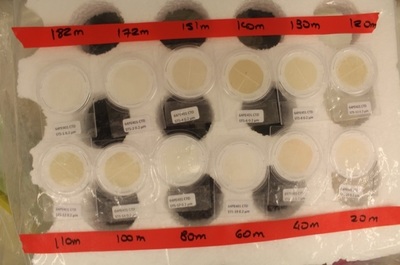
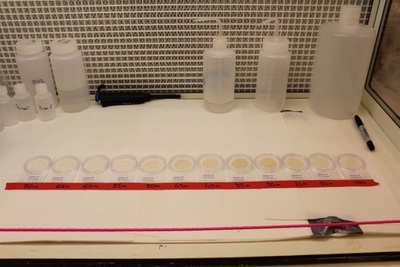
Although filling bottles can be slow, collecting the particles as samples can be really slow (it can take up to 5 hours for one sample- thankfully we don’t have to watch). To collect the particles, we use “flat” filters that we put into a plastic filter holder that allows water flowing out of the bottle spigot to flow through the filter and then into a bucket. We use two sizes, 0.2 and 0.02 µm size pore filter and we use a bucket (it is where the bucket science moto makes all sense!) to measure the volume of seawater that we filtered to allow us to get the concentrations of trace metals. For some of the filters, we have seen some small copepods (tiny animals about the size of a grain of sugar).
Depending on the oxygen concentration in the water column we process two different ways to process the sample. If the oxygen concentration is below 60 µmol/kg, we consider the sample anoxic, meaning that for a full preservation of the particles, we should work into a lab without air, which is obviously not possible. We therefore use a glove-bag that we fill with nitrogen gas. Once the sample is ready, we close valves up and below the filter holder to avoid any leakage of air into the sample, and we take it to the lab container and place them into the glove-bag. We remove the filters from the filter holder and place them into the Petri slides which are placed into an aluminium bag filled with nitrogen gas. We seal the bag and store the sample at - 20 °C for future analysis.
For oxic sample, with an oxygen concentration above 60 µmol/kg, we take the sample to a clean environment, under a HEPA hood and place the filter into double bagged Petri slides and hop freezer.
Depending on the oxygen concentration in the water column we process two different ways to process the sample. If the oxygen concentration is below 60 µmol/kg, we consider the sample anoxic, meaning that for a full preservation of the particles, we should work into a lab without air, which is obviously not possible. We therefore use a glove-bag that we fill with nitrogen gas. Once the sample is ready, we close valves up and below the filter holder to avoid any leakage of air into the sample, and we take it to the lab container and place them into the glove-bag. We remove the filters from the filter holder and place them into the Petri slides which are placed into an aluminium bag filled with nitrogen gas. We seal the bag and store the sample at - 20 °C for future analysis.
For oxic sample, with an oxygen concentration above 60 µmol/kg, we take the sample to a clean environment, under a HEPA hood and place the filter into double bagged Petri slides and hop freezer.
Time to go back to the laboratory on land and analyse the samples!
Cheers,
Marie
Cheers,
Marie





 RSS Feed
RSS Feed
